Development Technology of Synbiotic Yogurt With Cholesterol Lowering Ability
Economy
Sarengaole1,2, Ch.Tsend-Ayush1, Bayinjirigala3, Monhebilige2
1
Food Processing Technology of Mongolian University of
Science and Technology, Mongolia.
2
Key Laboratory of Dairy
Biotechnology and Engineering, Ministry of Education, China; Inner Mongolia
Agricultural University, Inner Mongolia, China.
3 College of Animal Science; Inner Mongolia Agricultural University, Inner Mongolia, China.
Abstract - The aim of this study is to determine the development technology of synbiotic yogurt with cholesterol lowering ability. The strains of lactic acid bacteria with cholesterol lowering ability was used as the stater cultures of AM2-6 and BM2-5. Inulin and Fructooligosaccharides was used for prebiotic combination. The most desirable conditions according to the Box-Behnken design was 1.77:1 of inoculation rate, 5 g/100g of inoculum, under 41.92℃ fermentation temperature in the condition 1.0% inulin, at the prediction of 98.0%. And 1.89:1 of inoculation rate, 5 g/100g of inoculum, under 42.87℃ fermentation temperature in the condition 1.5% FOS, repectively, at the prediction of 82.3%. The results of the orthogonal experiment by Box-Behnken design were consistent with the optimal condition results of single factor experiment in the actual operation which the inoculation rate of 2:1, 5 g/100g of inoculum and fermentation under 43℃.
Key words:
Cholesterol lowering ability; Synbiotic yogurt; development technology; Inulin; Fructooligosaccharides; Box-Behnken design.
Introduction
Commercial starter cultures was usually directly inoculated into foods
and beverages in industries (Sandine, 1996). At least one functional property
contributed to improving food quality in fermentation processes and benefited
to producing safety and healthy end products were characterized in the starter
cultures (Leroy et al, 2004).
In the recent years, more and more researches on the use of native
microorganism isolated from traditional dairy products for natural stater
cultures were studied (Peighambardousta et al, 2011; Qinglong et al, 2014).
Lactic acid bacteria played an important role in the use of natural stater
cultures in fermentation processes of foods and beverages (Jan et al, 2002).
Probiotics are viable microorganism that are beneficial to consumers
when ingested into foods and beverages with appropriate quantities (Graham, 2007). Lactic acid bacteria have been
used as probiotics (Lactobacilli, Streptococci,
Enterococci, Lactococci, Bifidobacteria)
over the years (Giuseppe B. et al, 2017; Antonia T. et al, 2017). Health advantages associated by probiotics are
improvement of lactose intolerance, increase in humoral immune responses,
biotransformation of isoflavone phytoestrogen to improve post-menopausal
symptoms, bioconversion of bioactive peptides for antihypertension, improvement
of serum lipid profiles, increase in natural resistance to infectious disease
in gastrointestinal tract, suppression of cancer, lower the cholesterol level
and improved digestion (Gibson G.R. et al, 1995; Nelly R. V. et al, 2013; Batmunkh M. et al, 2016;
Bira N. 2010).
Cholesterol is an organic molecule, a type of lipid. it is
biosynthesized by cells and is an essential structural component of cell
membranes. It is also the steroid hormones for biosynthesis of bile acid and vitamin D. Hepatic cells produce the
greatest amounts (Hanukoglu I., 1992). In general, two-thirds of the
cholesterol in the body is endogenously synthesized, and one-third is
exogenously ingested. Synthesis within the body starts with the mevalonate
pathway by the enzyme 3- hydroxy-3- methylglutaryl CoA (HMG-CoA) reductase and
finally synthesized in the body under a series of oxidases action (Wang W.,
2009). Exogenous intake of cholesterol is mainly obtained from the diet
and current foods are rich in lipids. Thus, a higher intake from food leads to
a net decrease in endogenous production and the increase of cholesterol in the
blood. Higher cholesterol in blood is associated with risks of heart disease
(Grundy et al, 1982; Pyorala, 1987; Gurr, 1992).
Numerous studies about lactic acid bacteria as probiotics with
cholesterol lowering ability were occurred in the recent years, especially in
various types of characteristic dairy products (Graham et al, 2006). According to the current
researches on probiotic lactic acid bacteria, two theories support that to
lower cholesterol. One theory is that some in
vitro studies showed the cells of lactic acid bacteria can absorb
cholesterol when grow in the high cholesterol medium containing bile salts
under anaerobic conditions, and reduce the cholesterol content in the medium
(Noh et al, 1997). One theory is that some in
vitro studies showed the bile salt hydrolase which produced by lactic acid
bacteria can deconjugate
combined bile
acid into free bile acid, then the free bile acid combines with cholesterol
into a complex to co-precipitate to lower the cholesterol content (Corzo et al,
1999; Pereira et al, 2003).
The definition of prebiotics are the non-digestible food ingredients
that benefit the host through selectively stimulate the growth and/or activity
of bacterial species already present in the colon (Gibson et al, 1995). Thus,
the effect of prebiotics are neither be hydrolysed nor absorbed in the upper
part of the gastrointestinal tract, and
selective for bacteria on the colon. A decreasing trend is showed in the
numbers of probiotics in human gut with age (Mitsuoka, 1992). To maintain a high level of probiotics, two
major strategies have been proposed. To continuously
intake foods containing probiotics or supply foods containing prebiotics (Gomes
et al, 1999).
Oligosaccharides are the most widely researched prebiotics. It consisting of 2 to 20 sugar units and some occur naturally in foods such as leek, asparagus, chicory, garlic, onion and soybean. There are numerous properties of oligosaccharides which serve the desired attributes in prebiotics. High selectivity and efficiency in metabolism by bifidobacteria and lactobacilli have enabled the supplementation of prebiotics at low dosage (Gibson G. R. et al 2000).
Inulin and Fructooligosaccharides (FOS) are the most studied
oligosaccharides for their prebiotic properties. Inulin type fructans contain
both glucosyl and fructosyl molecules, the number of fructose units are varying
from 2 to 70. A rich source of inulin naturally distributed in the plants. It
contains an unique β(2→1) bonds that prevent inulin from being digested by
carbohydrate so that reduce the caloric
value and increase the dietary fibre effects (S. S. R. Ali et al 2016). The IUPAC/IUB JCBN and the AOAC defined FOS as
the fructose oligosaccharide containing 2 to 10 monosaccharide residues linked
by glycosidic bonds. Similar to inulin, β(2→1) bonds are also inside FOS and it
contributes to resistance towards hydrolysis by human digestive enzymes. Thus,
inulin and FOS can pass through the mouth, stomach and small intestine without
being metabolized (C. F. Balthazar
et al, 2017).
In the previous studies, almost all of the inulin and FOS ingested
enters the colon and is completely fermented by colonic microbiota. All these
fermentation produced energy, short
chain fatty acids and lactic acid. The increase in lactic acid concentrations,
decrease the luminal pH to inhibit putrefactive microorganism (Niness, 1999).
Combination of probiotics and prebiotics is called synbiotics. It has synergistic effects that not only promoting growth of the bacteria in the colon, but also improving the survival, implantation and growth of newly added probiotic strains. In the present study, inulin and FOS will be combined with the probiotic Lactic acid bacteria strains for the use in development technology of synbiotic products with cholesterol lowering ability (Oliveira et al, 2009a).
Development technology of synbiotic yogurt with cholesterol lowering ability will be produced under the fermentation process of stirred yogurt production with some modification. The strains of lactic acid bacteria with cholesterol lowering ability was provided by Key Laboratory of Dairy Biotechnology and Engineering, Ministry of Education, China will be used as the stater cultures. Inulin and FOS will be used for prebiotic combination. The combination of probiotic starter cultures and prebiotics will be optimized by the determination of their cholesterol lowering ability. Optimal inoculum and rate of starter cultures, optimal fermentation period and temperature will be selected as the parameter to be confirmed in the development technology of synbiotic yogurt with cholesterol lowering ability through the determination of pH, titradable acidity and viable count.
Material and Methods
1
Milk preparation
Skim milk powder (Anchor, New Zealand) was pretreated and standardized
according to GB 19644-2010 and GB 19301-2010. The quality inspection of
hydrated milk was determined according to GB 19644-2010.
2 Viable
count in
prebiotic conditions
Freeze dried cells of the Lb.
plantarum strains AM2-6 and BM2-5 with cholesterol lowering ability were used as the production starter
cultures for the development technology of synbiotic yogurt (provided by Key
Laboratory of Dairy Biotechnology and Engineering, Ministry of Education,
China). It was incubated under different
concentration of prebiotic
inulin and FOS according to the method by
S. S. R. Ali
et al, 2016. The viable count of typical strains in the presence of different
prebiotic concentrations was determined. The inoculation of prebiotic was under
the national standard of GBT 23528-2009.
3 Cholesterol lowering ability
on synbiotic effect
The cholesterol lowering ability of each freeze dried strain cells in
prebiotic condition was determined according to their cholesterol lowering
ability. The cholesterol lowering ability was
determined
using o-phthalaldehyde method as
described by Rudel L. L. et al, 1973 and M.G. Shehata et al, 2016. The
experiment of evaluation of cholesterol lowering ability was replicated twice
and in comparison with the standard strain Lb.
plantarum NBRC 15891T. The optimal addition of prebiotic inulin
and FOS was obtained according to their cholesterol lowering ability.
4 Determination of optimal
rate of starter cultures
The production process of synbiotic yogurt with cholesterol lowering
ability was based on the traditional production process of stirred yogurt with
some modification according to GB 19302-2010. Skim milk power was used for milk
preparation. The inoculation rate of starter cultures was different in
prebiotic condition. The pH value, titratable acidity and viable count of each
yogurt was determined.
5 Determination of optimal
inoculum of starter cultures
The synbiotic yogurt with cholesterol lowering ability was produced as
mentioned above. The starter cultures was inoculated at different inoculum in
prebiotic condition.
6 Determination of optimal
fermentation period and temperature
The synbiotic yogurt with cholesterol lowering ability was produced
under different temperature in prebiotic
condition as mentioned above.
7 Box-Behnken design of
synbiotic yogurt with cholesterol lowering ability
The optimal production process of the synbiotic yogurt with cholesterol
lowering ability was obtained through response surface experiment design. Box
Behnken Design was performed using the Design-Expert 8.0.6 software.
8 Physical and chemical
analysis of synbiotic yogurt with cholesterol lowering ability
The production process under the optimal fermentation conditions of
synbiotic yogurt with cholesterol lowering ability was obtained. The physical
and chemical analysis of protein, fat and ash was determined according to GB
5009. 6-2016, GB 5009. 5-2016, and GB 5009. 4-2016, respectively. The total
solid content was determined according to GB 5413. 39-2010.
Amino acid were determined using L-8900 amino acid
analyzer according to GB 5009. 124-2016. Concentrations of vitamins A and E
were determined by RP-HPLC according to GB 5009. 82-2016. Concentrations of
vitamins B1 were determined by RP-HPLC according to GB 5009.
84-2016. Concentrations of vitamins B2 were determined by HPLC
according to GB 5009. 85-2016. Concentrations of vitamins C were
determined by HPLC according to GB 5009. 86-2016.
9 Sensory evaluation of
synbiotic yogurt with cholesterol lowering ability
Sensory
evaluation was performed by seven trained researchers according to RHB
103-2004.
10 Storage of synbiotic yogurt
with cholesterol lowering ability
The synbiotic yogurt with cholesterol lowering ability was refrigerated and stored under 4℃ for 21 days (GB 19302-2010). The pH, titratable acidity and viable count was determined in every 7 days.
Results and Discussions
1 Viable
count in
prebiotic conditions
Freeze dried cells of the Lb. plantarum strains AM2-6 and BM2-5 were incubated anaerobically under 37℃ for 24 h on the freshly prepared MRS broth. After 20 h of culturing, the MRS broth was centrifuged, and the cell pellet washed twice by D. W.. The supernatant was removed and 0.1 M PBS solutions (pH 6.8) was added. The mixture was vortexed, and freeze dried under -20℃ for 48 h. The prebiotic inulin and FOS was prepared according to the method by S. S. R. Ali et al, 2016 in different concentration of 0.5%, 1%, 1.5% (w/v). Each freeze dried typical LAB strain cells were incubated anaerobically in different prebiotic conditions under 37℃ for 24 h. The viable count was determined at the end of the incubation period (Table 1). The addition of prebiotic inulin and FOS, didn’t show a significant effect on the viable count (P>0.05). A similar result was also reported by Tatdao P., et al (2009).
Table
1. Viable count of strains AM2-6 and BM2-5 in the prebiotic conditions.
![]()
Viable Count Inulin (%) FOS (%)
|
Log 10 CFU/g |
0 |
0.5 |
1.0 |
1.5 |
0 |
0.5 |
1.0 |
1.5 |
|
AM2-6 |
7.39 |
7.36 |
7.33 |
7.36 |
7.42 |
7.31 |
7.41 |
7.33 |
BM2-5 7.36 7.33 7.38 7.38 7.40 7.32 7.35 7.36
All data was repeated twice and recorded as mean±SD.
1
Cholesterol lowering ability on synbiotic effect
The Lb. plantarum strains
AM2-6 and BM2-5 showed a certain cholesterol lowering ability in the prebiotic
conditions, as shown in Table 2.
Under the condition of prebiotics, the two probiotics did not lose their
cholesterol lowering ability. Although in the conditions of 0.5%, 1.0% and 1.5%
of inulin condition, the cholesterol lowering rate was lower than the single
strains in blank condition. Thus, it could be considered that the two strains
had a certain degree of cholesterol lowering ability, but both of their
cholesterol lowering ability was decreased when collaborate with inulin. The
cholesterol lowering ability of the two strains under the condition of 1.0%
inulin is close to the blank one. The cholesterol lowering rate of AM2-6 was
32.36% and BM2-5 was 27.23%. Therefore, the optimal synbiotic effect on the two
strains was in the condition of 1.0% inulin.
Two strains showed an increased cholesterol lowering rate in the
condition of 1.5% FOS. It suggested that, under the condition of 1.5% FOS, the
two LAB strains were more effective on lowering cholesterol. The cholesterol
lowering rate of AM2-6 was 38.31% and BM2-5 was 29.74% in the condition of 1.5%
FOS. The optimal synbiotic effect on the two strains was in the condition of
1.5% FOS.
The synbiotic effect on probiotics showed different fermentation ability, this result was supported by Kaplan et al (2000) who reported that several strains of lactic acid bacteria were able to ferment fructooligosaccharides well, but not all the strains.
Table 2. Cholesterol lowering ability of strains AM2-6 and
BM2-5 in the prebiotic conditions.
|
Strain |
Lowering Cholesterol |
AM2-6 Cholesterol Lowering Rate |
Lowering Cholesterol |
BM2-5 Cholesterol Lowering Rate |
|
|
(μmol/ml) |
(%) |
(μmol/ml) |
(%) |
|
Inulin (%) 0 |
48.56±1.27 |
32.37 |
43.47±3.34 |
28.98 |
|
0.5 |
45.68±0.37 |
30.72 |
41.96±1.13 |
27.01 |
|
1.0 |
47.32±1.23 |
32.36 |
42.19±2.03 |
27.23 |
|
1.5 |
46.96±0.68 |
31.02 |
41.17±1.13 |
26.21 |
|
FOS (%) 0 |
48.56±1.27 |
32.37 |
43.47±3.34 |
28.98 |
|
0.5 |
51.96±1.17 |
37.03 |
42.87±0.69 |
27.91 |
|
1.0 |
52.39±2.12 |
37.43 |
43.39±0.48 |
28.43 |
|
1.5 |
53.27±1.18 |
38.31 |
44.64±1.32 |
29.68 |
All data was repeated twice and recorded as mean±SD.
1
Determination of optimal rate of starter cultures
The synbiotic yogurt with cholesterol lowering ability was produced
using the production starter cultures of AM2-6 and BM2-5. The production
process of synbiotic yogurt with cholesterol lowering ability was based on the
traditional production process of stirred yogurt according to GB 19302-2010
with some modification. To confirm the optimal production process, the optimal
rate, inoculum and temperature were determined as above. The flow chart of
development technology of synbiotic yogurt with cholesterol lowering ability
was shown as Fig 1.
Inulin was widely used as the fat substitute in yogurt production
processes. It forming a creamy structure, after mixing into the water (Li Y. et al, 2015). Therefore, skim milk
powder was used for the production process of synbiotic yogurt with cholesterol
lowering ability. Briefly, skim milk powder was 11.5% (w/v) hydrated with
addition of 1.0% inulin and 1.5% FOS respectively (Li Y. et al, 2015). Then heated under 95℃ for 5 min. After rapidly
cooled to 43℃, the production starter
cultures was inoculated at the
rate of 1:1, 2:1, 1:2, respectively (Li H. et al, 2008). The inoculum was 3
g/100g of raw milk and fermentation temperature was under 43℃ according to the
result in section 3.6. The pH value,
titratable acidity was shown in Fig 2, viable count and sensory evaluation
score was shown in Table 3.
According to the fermentation ability of synbiotic yogurt with 1:1, 1:2, 2:1 rate of starter cultures in the conditions of 1.0% inulin and 1.5% FOS, AM2-6: BM2-5 at the rate of 2:1 showed a stronger fermentation ability (Fig 2). In the conditions of both 1.0% inulin and 1.5% FOS, the fermentation period was about 6 h (pH 4.6). Combine the result of viable count and sensory score, strain rate AM2-6: BM2-5 of 2:1 was considered as the optimal rate of the mixed starter cultures.
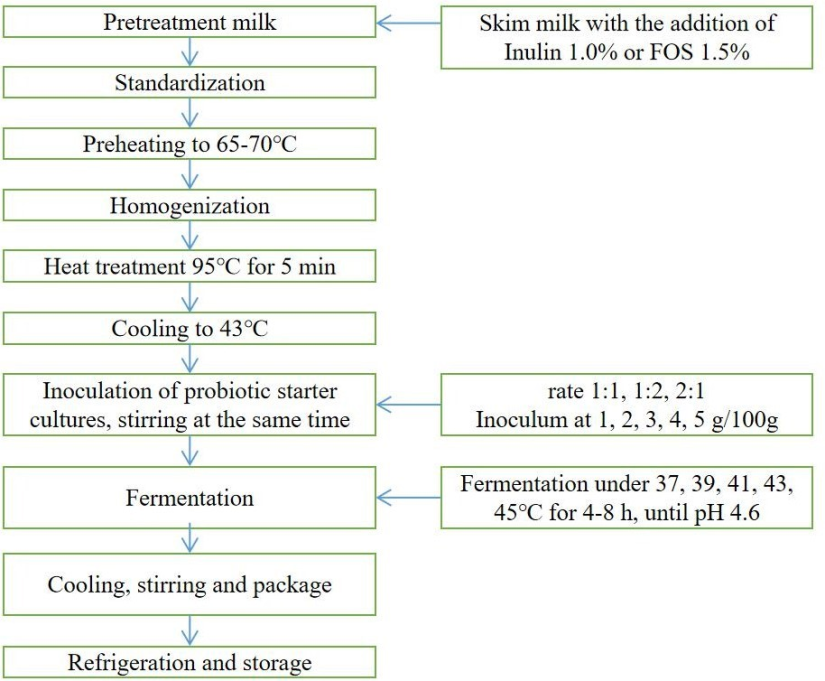
Fig 1. The flow chart of development technology of synbiotic yogurt with cholesterol lowering ability.
Fig 2. The fermentation ability of synbiotic yogurt with 1:1, 1:2, 2:1 rate of starter cultures in the conditions of 1.0% inulin and 1.5% FOS. A: pH measurement. B: Titratable acidity.
Table 3. Effect of different rate of starter cultures on the yogurt in the conditions of 1.0% inulin and 1.5% FOS.

5
Determination of optimal inoculum of starter cultures
Skim milk powder was used for the production process of synbiotic
yogurt with cholesterol lowering ability as mentioned above. The inoculation
rate of starter cultures was 1:1 and the fermentation temperature was 43℃. The
starter cultures was inoculated at 1 g/100g, 3 g/100g and 5 g/100g of inoculum
in the condition of 1.0% inulin and 1.5% FOS. The pH value, titratable acidity
was shown in Fig 3, viable count and sensory evaluation score was shown in
Table 4.
According to the fermentation ability of synbiotic yogurt at different inoculum of starter cultures, 5% of inoculum was considered as the optimal inoculum in the conditions of both 1.0% inulin and 1.5% FOS (Fig 3). The fermentation period was about 6 h (pH 4.6).
Fig 3. The fermentation ability of synbiotic yogurt at 1%, 3% and 5% of inoculum of starter cultures in the conditions of 1.0% inulin and 1.5% FOS. A: pH measurement. B: Titratable acidity.
Table
4. Effect of different inoculum of starter cultures on the yogurt in the
conditions of 1.0% inulin and 1.5% FOS.
6 Determination of optimal fermentation period and temperature
Skim milk powder was used for the production process of synbiotic
yogurt with cholesterol lowering ability as mentioned above. The starter
cultures was inoculated at the rate of 1:1 and 3 g/100g of inoculum in the
condition of 1.0% inulin and 1.5% FOS. The fermentation temperature was 37℃, 39℃, 41℃, 43℃ and 45℃. The
fermentation period was indicated by pH and titratable acidity (Fig 4), viable
count and sensory evaluation score was shown in Table 5.
According to
the fermentation ability of synbiotic yogurt under different fermentation temperature,
43℃ was considered as the optimal temperature in the conditions of both 1.0%
inulin and 1.5% FOS (Fig 4). The fermentation period was about 6 h (pH 4.6).
Compared to cholesterol lowering yogurt without prebiotic condition, the fermentation period of synbiotic yogurt with cholesterol lowering ability required more time to the end of the fermentation. The best fermentation observed under fermentation temperature under 43℃ and the fermentation period was about 6h.
Table
5. Effect of different fermentation temperature on the yogurt in the conditions
of 1.0% inulin and 1.5% FOS.
7 Box-Behnken design of synbiotic yogurt with cholesterol lowering ability
In Box-Behnken design of the optimal production process of synbiotic
yogurt with cholesterol lowering ability, 3 numeric factors and 3 responses
were set. The 3 numeric factors were inoculation rate of strain AM2-6 and
BM2-5, the inoculum and fermentation temperature (Table 6). And 3 responses were pH, titradable acidity and the
viable count of each produced yogurt (Table
7). Two sets of designs were
made under the conditions of 1.0% inulin and 1.5% FOS, respectively.
According to the Box-Behnken design, the final equation in terms of
coded factors was performed as the following formula in the conditions of 1.0%
inulin:
R1 = +4.18950 +0.015000 * X1 -0.011250 * X2
+0.010938 * X3。
R2
= +78.82 -0.62 * X1 +1.13 * X2 -2.25 * X3.
R3
= +7.08 +0.010 * X1 +0.091 * X2 -0.041 * X3
+7.500E-003 * X1 * X2 +0.067 * X1 * X3
+0.11 * X2
* X3.
The final equation in terms of coded factors was performed as the following formula in the conditions of 1.5% FOS:
R2 = +68.40 +3.38 * X1 +2.50 * X2 +1.88 * X3 +1.00 * X1 * X2 +0.25 * X1 * X3 -1.00 * X2 * X3 +3.42* X 2 +2.17 * X 2 +0.93 * X 2.
R3 = +7.08 +0.056 * X1 +0.056 * X2 +0.10 * X3 +0.000 * X1 * X2 +0.038 * X1 * X3 -0.013 * X2 * X3-0.012 * X 2 +0.028 * X 2 -0.11 * X 2.
The effect of inoculation rate, inoculum and fermentation temperature on the pH, titradable acidity and viable count which designed by Box-Behnken design of the optimal production process of synbiotic yogurt with cholesterol lowering ability in the conditions of 1.0% inulin and 1.5% FOS. The most desirable conditions combined the numeric factors on the responses according to the Box-Behnken design was 1.77:1 of inoculation rate, 5 g/100g of inoculum, under 41.92℃ fermentation temperature in the condition 1.0% inulin, at the prediction of 98.0%. And 1.89:1 of inoculation rate, 5 g/100g of inoculum, under 42.87℃ fermentation temperature in the condition 1.5% FOS, repectively, at the prediction of 82.3%. The results of the orthogonal experiment by Box-Behnken design were consistent with the optimal condition results of single factor experiment in the actual operation which the inoculation rate of 2:1, 5 g/100g of inoculum and fermentation under 43℃. The most desirable conditions of development technology of synbiotic yogurt with cholesterol lowering ability by Box-Behnken design was shown as Fig 5.
Table 6. The numeric factors of synbiotic yogurt with cholesterol lowering ability.
Fig 5. The most desirable conditions of development technology of synbiotic yogurt with cholesterol lowering ability. A: 1% inulin; B: 1.5% FOS.
7 Physical and chemical
analysis of synbiotic yogurt with cholesterol lowering ability
The optimal production process of synbiotic yogurt with cholesterol
lowering ability was inoculation rate of 2:1, 5 g/100g of inoculum and
fermentation under 43℃. And the fermentation
period was about 6 h.
Physical and chemical index was determined as shown in Table 8. The
standard strain Lb. plantarum NBRC
15891T was prepared as the production starter cultures for
comparison. All data was within the standards by national standards GB
19302-2010.
Amino acid content was shown in Table 9. According to the propose by
WHO/FAO, the cholesterol lowering yogurt reached the standard and it could be
considered as a resource of good amino acid. And the
concentrations of vitamins
A, E, B1, B2, C were determined as shown in Table 10.
Table 8. The physical and chemical index of synbiotic yogurt with cholesterol lowering ability.
*AA: Amino Acid; NEAA: Non-Essential Amino Acid; SEAA: Semi-Essential Amino Acid;
EAA:Essential Amino Acid; TAA: Total Amino Acid.
All data was repeated twice and recorded as mean±SD.
Table 10. The vitamins content of A, E, B1, B2 and C of synbiotic yogurt with cholesterol lowering ability.
8 Storage of synbiotic yogurt
with cholesterol lowering ability
The synbiotic yogurt with cholesterol lowering ability was
refrigerated and stored under 4℃ for 21
days. The pH and titratable acidity was determined in every 7 days (Table 11). During the storage of
synbiotic yogurt with cholesterol lowering ability,
the number of viable count, acidity was decreased slightly as the same
with cholesterol lowering yogurt.
Regardless of the level of inulin and FOS, the protective effect of prebiotics on the survival and viability of LAB has not been verified in the synbiotic yogurt (Hernandez H. et al, 2012). All the conditions of inulin and FOS confirmed the adequacy for bacteria growth in the synbiotic yogurt. The viable count has established 7 log cfu/g as a sum of LAB in yogurt and were in accordance with Codex Alimentarius (2010).
Table 11.
The pH, titratable acidity and viable count of synbiotic yogurt with
cholesterol lowering ability during 21 days of storage.
Conclusions
The Lb. plantarum strains
AM2-6 and BM2-5 showed a certain cholesterol lowering ability in the prebiotic
conditions. Although in the conditions of 0.5%, 1.0% and 1.5% of inulin
condition, the two strains had a certain degree of cholesterol lowering
ability, but both of their cholesterol lowering ability was decreased when
collaborate with inulin. The cholesterol lowering ability of the two strains under
the condition of 1.0% inulin is close to that in the blank condition. The
cholesterol lowering rate of AM2-6 was 32.36% and BM2-5 was 27.23%. The optimal
synbiotic effect on the two strains was in the condition of 1.0% inulin. Two strains showed an increased
cholesterol lowering rate in the condition of 1.5% FOS. The cholesterol
lowering rate of AM2-6 was 38.31% and BM2-5 was 29.74% in the condition of 1.5%
FOS. The optimal synbiotic effect on the two strains was in the condition of
1.5% FOS.
The synbiotic yogurt with cholesterol lowering ability was produced
using the production starter cultures of AM2-6 and BM2-5. The most desirable
conditions combined the numeric factors on the responses according to the
Box-Behnken design was 1.77:1 of inoculation rate, 5 g/100g of inoculum, under
41.92℃ fermentation temperature in the condition 1.0% inulin, at the prediction
of 98.0%. And 1.89:1 of inoculation
rate, 5 g/100g of inoculum, under 42.87℃ fermentation temperature in the condition 1.5% FOS, repectively, at the
prediction of 82.3%. The results of the orthogonal experiment by Box-Behnken
design were consistent with the optimal condition results of single factor
experiment in the actual operation which the inoculation rate of 2:1, 5 g/100g
of inoculum and fermentation under 43℃.
Acknowledgment
Specially grateful to the professor Menghebilige provided the relevant experimental conditions in the course of the completion of the present study.
References
Antonia T.,
Angelika L. G., Loulouda B., Maria K., Athanasios A. K., Guillermo R. C. (2017)
Novel cheese production by incorporation of sea buckthorn berries (Hippophae
rhamnoides L.) supported probiotic cells. LWT - Food Science and Technology 79:
616-624
Batmunkh M.,
Batjargal B. (2016) Characteristics of Probiotics Lactic Acid Bacteria.
Student's Academic Papers: 204-210 [Original in Mongolia: М. Батмөнх, Б.
Батжаргал (2016) Зарим Сүүн Хүчлийн Бактерийн Пробиотик Шинж Чанарын Судалгаа.
Оюутны Эрдэм Шинжилгээний Илтгэлийн Эмхэтгэл: 204-210]
Bira N. (2010) Food therapy for cancer
prevention, the task of prebiotics and probiotics. Diagnosis
(046)
[Original in Mongolia: Бира Н. (2010) Хавдраас Урьдчилан Сэргийлэхэд Хүнсний
Эслэг, Пребиотик Ба Пробиотикийн Үүрэг. Онош (046)]
CF. Balthazar, H. L. A. Silva,
A. H.
Vieira, R. P. C. Neto, L. P. Cappato, P.
T.
Coimbra, J. Moraes, M.
M. Andrade, V. M. A. Calado, D. Granato, M. Q.
Freitas, M. I. B. Tavares, R. S. L. Raices, M. C. Silva,
A. G. Cruz
(2017) Assessing the effects of different prebiotic dietary oligosaccharides in
sheep milk ice cream. Food Research International 91: 38-46
Corzo G., Gilliland S. E. (1999)
Measurement of bile salt hydrolase activity from Lactobacillus acidophilus based on disappearance of conjugated of
bile salts. Journal of Dairy Science
82: 466-471 Gibson G. R., Otaway P. B., Robert A. R. (2000) Prebiotics: New
developments in functional foods. London: Chandos Publishing (Oxford) Limited
Gibson G. R., Roberfroid M. B.
(1995) Dietary modulation of the human colonic microbiota:
introducing the concept of prebiotics. J Nutr 125: 1401-1412
Giuseppe B., Nicoletta M., Alessandro D. C., Mariantonietta S.,
Raffaele C., Maria A. (2017)
Commercially
standardized process for probiotic “Italico” cheese production. LWT - Food Science
and Technology 79: 601-608
Gomes A. M. P.
and Malcata F. X. (1999) Bifidobacterium spp. and Lactobacillus acidophilus:
biological, biochemical, technology and therapeutical properties relevant to
use as probiotics. Trend Food Sci Technol 10: 139-157
Graham F.,
Roostita B. (2006) Yeasts in Foods and Beverages: The public health and
probiotic significance. Springer Berlin Heidelberg: 381-397
Graham H. F. (2007) Yeasts in Foods and Beverages: Impact
on product quality and safety. Current Opinion in Biotechnology 18: 170-175
Grundy S. M.,
Brheimer D., Blackburn H., Brown W. V., Kwiterovich
P. O., Mattson F., Schonfeld G., Weidman W. H., (1982)
Rational of the diet-heart statement of the American Heart Association
report of the Nutrition Committee,
Circulation 65: 839A-854A
Gurr M. I.
(1992) Dietary lipids and coronary disease: Old evidence, new perspective,
Progress in Lipid Research 31: 195-243
Hanukoglu I.
(1992) Steroidogenic enzymes: structure, function, and role in regulation of
steroid hormone biosynthesis. J Steroid Biochem Mol Biol. 43(8): 779-804
Jan T. M. W.,
Eman H. E. A., Jeroen H., Gerrit S. (2002) Microbes from raw milk for fermented
dairy products. International Dairy Journal 12: 91-109
Kaplan, H.,
Hutkins, R. W. (2000) Fermentation of fructooligosaccharides by lactic acid
bacteria and bifidobacteria. Applied and Environmental Microbiology 66:
2682-2684
Leroy F., de
V. L. (2004) Lactic acid bacteria as functional starter cultures for the food
fermentation industry. Trends in Food Science & Technology 15: 67-78
Li H., Liu H. (2008) Optimization of the development technology od
fermented yogurt production process. Modern Food Science and Technology 24: 4
[Original in Chinese: 李宏梁,
刘辉 (2008)
发酵酸奶生产工艺优化.
现代食品科技 24:
4]
Li Y., Liu L., Tong L., Sun J., Song L. (2015) Effect of
inulin on the quality of low sugar, low fat yogurt. Food and Fermentation Industrries 41 (12): 131-134 [Original in Chinese: 李雨露,刘丽萍, 佟丽媛,孙建华,宋立. (2015) 菊粉对低糖低脂酸奶品质的影响. 食品与发酵工业 41 (12):
131-134]
M.G. Shehata,
S.A. El Sohaimy, Malak A. El-Sahn, M.M. Youssef (2016) Screening of isolated
potential probiotic lactic acid bacteria for cholesterol lowering property and
bile salt hydrolase activity. Annals of Agricultural Science 61(1): 65-75
Mitsuoka T. (1992) Intestinal flora and
aging. Nutr Rev 50: 438-446
Nelly R. V., Niklas T., Pernilla L., Christine A K., Rolf C., Magnus D., Carina Ö.,
Anne CR T., Olle H., Ingegerd J.
(2013) Characterization and in vitro properties of oral lactobacilli in
breastfed infants. BMC Microbiology 13:193
Niness K. R.
(1999) Nutritional and health benefits of inulin and oligofructose. J Nutr 129:
1402S-1406S
Noh D. O., Kim
S. H., Gilliland S. E., (1997) Incorporation of cholesterol into the cellular
membrane of Lactobacillus acidophilus ATCC 43121,
Journal of Dairy Science 80: 3107-3113
Oliveira R. P.
D. S., Perego P. C. A., De Oliveira M. N. (2009a) Growth and acidification
performance of probiotics in pure culture and co-culture with Streptococcus
thermophilus: The effect of inulin. LWT-Food Science and Technology.
42:1015-1021
Peighambardousta
S. H.,Golshan T. A., Hesari J. (2011) Application of spray drying for
preservation of lactic acid starter cultures: A review. Trends in Food Science
& Technology 22: 215-224
Pereira D. I.
A., McCartney A. L., Gibson G. R., (2003) An in vitro study of the probiotic potential of a
bile-salt-hydrolyzing Lactobacillus
fermentum strain, and determination of its cholesterol-lowering properties,
Applied and Environmental Microbiology 69: 4743-4752
Pyorala K.
(1987) Dietary cholesterol in relation to plasma cholesterol and coronary heart
disease. The American Journal of Clinical Nutrition 45: 1176-1184
Qinglong W., Christine K. W., Cheung, Nagendra P. S.
(2014) Towards galactose
accumulation in dairy foods fermented by conventional starter cultures:
Challenges and strategies. Trends in Food Science & Technology xx: 1-13
Rudel L. L.,
Morris M. D. (1973) Determination of cholesterol using o-phtaldealdehyde. J
Lipid Res. 14: 364-366
Sandine W. E.
(1996) Dairy starter cultures: Commercial production of dairy starter cultures.
Wiley-VCH. New York, USA: 191-206.
SS. R. Ali, K.
Ambasankar, S. Nandakumar, P. E. Praveena, J. Syamadayal (2016) Effect of
dietary prebiotic inulin on growth, body composition and gut microbiota of
Asian seabass (Lates calcarifer). Animal Feed Science and Technology 217: 87-94
Tatdao P., Frank S. (2009)
Probiotic stability of yoghurts containing Jerusalem artichoke inulins during
refrigerated storage. JOURNAL OF FUNCTIONAL FOODS 1: 311-318
Wang W. (2009) Screening
of probiotics with cholesterol-lowering function. Master’s thesis of
Heilongjiang University [Original
in Chinese: 王巍 (2009) 具有降胆固醇功能益生菌的筛选. 黑龙江大学硕士论文]
ATTACHMENT
GB 5009. 4-2016 National Food Safety Standard - Determination of Ash
in Food
GB 5009.
5-2016 National Food Safety Standard -
Determination of Protein in Food GB 5009. 6-2016 National Food Safety Standard
- Determination of Fat in Food
GB 5009. 82-2016 National Food
Safety Standard - Determination of Vitamin A, D, E in Food GB 5009. 84-2016
National Food Safety Standard - Determination of Vitamin B1 in Food
GB 5009. 85-2016 National Food
Safety Standard - Determination of Vitamin B2 in Food GB 5009. 86-2016 National
Food Safety Standard - Determination of Vitamin C in Food GB 5009. 124-2016
National Food Safety Standard - Determination of Amino Acid in Food GB 5413.
39-2010 National Food Safety Standard - Non-Fat Milk Solids
GB 19301-2010 National Food Safety Standard - Fresh Milk
GB 19302-2010 National Food
Safety Standard - Fermented milk GB 19644-2010 National Food Safety Standard -
Milk Powder
GBT 23528-2009 National Food
Safety Standard - Fructooligosaccharides RHB 103-2004 Evaluation Rules for
Sensory Quality of Yogurt
 Ulaanbaatar
Ulaanbaatar